Page 442 - Adsorption by Powders and Porous Solids
P. 442
ADSORPTION BY POWDERS AND POROUS SOLJDS
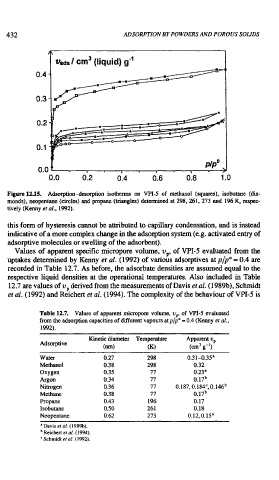
Figure 12.15. Adsorption-desorption isotherms on VPI-5 of methanol (squares), isobutane (dia-
monds), neopentane (circles) and propane (triangles) detennined at 298, 261, 273 and 196 K. respec-
tively (Kenny et al.. 1992).
this form of hysteresis cannot be attributed to capillary condensation, and is instead
indicative of a more complex change in the adsorption system (e.g. activated entry of
adsorptive molecules or swelling of the adsorbent).
Values of apparent specific micropore volume, v,, of VPI-5 evaluated from the
uptakes determined by Kenny et al. (1992) of various adsorptives at p/pO = 0.4 are
recorded in Table 12.7. As before, the adsorbate densities are assumed equal to the
respective liquid densities at the operational temperatures. Also included in Table
12.7 are values of v, derived from the measurements of Davis et al. (1989b), Schmidt
et al. (1992) and Reichert et al. (1994). The complexity of the behaviour of VPI-5 is
Table 12.7. Values of apparent micropore volume, v,, of VPI-5 evaluated
from the adsorption capacities of different vapours at p/p0 = 0.4 (Kenny et of.,
1992).
Kinetic diameter Temperature Apparent v,
Adsorptive
(nm) (K) (cm3 g-')
Water 0.27 298 0.31-0.35'
Methanol 0.38 298 0.32
Oxygen 0.35 77 0.23"
Argon 0.34 77 0.17~
Nitrogen 0.36 77 0.187,O. 184', 0.146
Methane 0.38 77 0.17~
Propane 0.43 196 0.17
Isobutane 0.50 261 0.18
Neopentane 0.62 273 O.12,0.1Sa
'Davis et a!. (1989b).
Reichen er 01. (1994).
'Schmidt et 01. (1992).