Page 319 - Fluid mechanics, heat transfer, and mass transfer
P. 319
SHELL AND TUBE HEAT EXCHANGERS
300
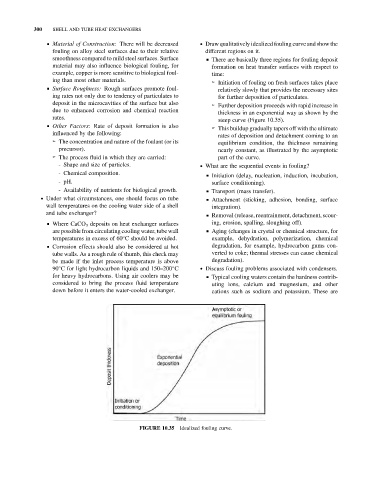
& Material of Construction: There will be decreased . Draw qualitatively idealized fouling curve and show the
fouling on alloy steel surfaces due to their relative different regions on it.
smoothness compared to mild steel surfaces. Surface & There are basically three regions for fouling deposit
material may also influence biological fouling, for formation on heat transfer surfaces with respect to
example, copper is more sensitive to biological foul- time:
ing than most other materials. ➢ Initiation of fouling on fresh surfaces takes place
& Surface Roughness: Rough surfaces promote foul-
relatively slowly that provides the necessary sites
ing rates not only due to tendency of particulates to for further deposition of particulates.
deposit in the microcavities of the surface but also ➢ Further deposition proceeds with rapid increase in
due to enhanced corrosion and chemical reaction
thickness in an exponential way as shown by the
rates.
steep curve (Figure 10.35).
& Other Factors: Rate of deposit formation is also
➢ This buildup gradually tapers off with the ultimate
influenced by the following:
rates of deposition and detachment coming to an
➢ The concentration and nature of the foulant (or its
equilibrium condition, the thickness remaining
precursor). nearly constant, as illustrated by the asymptotic
➢ The process fluid in which they are carried: part of the curve.
- Shape and size of particles. . What are the sequential events in fouling?
- Chemical composition. & Initiation (delay, nucleation, induction, incubation,
- pH. surface conditioning).
- Availability of nutrients for biological growth. & Transport (mass transfer).
. Under what circumstances, one should focus on tube & Attachment (sticking, adhesion, bonding, surface
wall temperatures on the cooling water side of a shell integration).
and tube exchanger? & Removal (release, reentrainment, detachment, scour-
& Where CaCO 3 deposits on heat exchanger surfaces ing, erosion, spalling, sloughing off).
are possible from circulating cooling water, tube wall & Aging (changes in crystal or chemical structure, for
temperatures in excess of 60 C should be avoided. example, dehydration, polymerization, chemical
& Corrosion effects should also be considered at hot degradation, for example, hydrocarbon gums con-
tube walls. As a rough rule of thumb, this check may verted to coke; thermal stresses can cause chemical
be made if the inlet process temperature is above degradation).
90 C for light hydrocarbon liquids and 150–200 C . Discuss fouling problems associated with condensers.
for heavy hydrocarbons. Using air coolers may be & Typical cooling waters contain the hardness contrib-
considered to bring the process fluid temperature uting ions, calcium and magnesium, and other
down before it enters the water-cooled exchanger. cations such as sodium and potassium. These are
FIGURE 10.35 Idealized fouling curve.