Page 430 - Fundamentals of Enhanced Oil and Gas Recovery
P. 430
418 Afshin Tatar
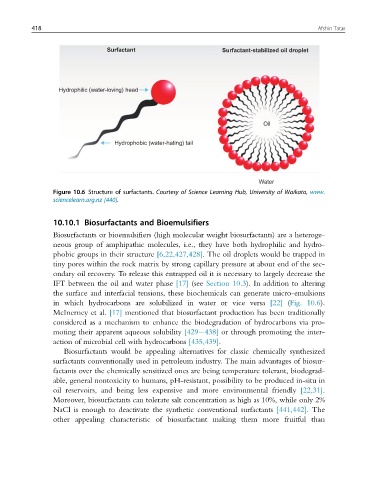
Surfactant Surfactant-stabilized oil droplet
Hydrophilic (water-loving) head
Oil
Hydrophobic (water-hating) tail
Water
Figure 10.6 Structure of surfactants. Courtesy of Science Learning Hub, University of Waikato, www.
sciencelearn.org.nz [440].
10.10.1 Biosurfactants and Bioemulsifiers
Biosurfactants or bioemulsifiers (high molecular weight biosurfactants) are a heteroge-
neous group of amphipathic molecules, i.e., they have both hydrophilic and hydro-
phobic groups in their structure [6,22,427,428]. The oil droplets would be trapped in
tiny pores within the rock matrix by strong capillary pressure at about end of the sec-
ondary oil recovery. To release this entrapped oil it is necessary to largely decrease the
IFT between the oil and water phase [17] (see Section 10.3). In addition to altering
the surface and interfacial tensions, these biochemicals can generate micro-emulsions
in which hydrocarbons are solubilized in water or vice versa [22] (Fig. 10.6).
McInerney et al. [17] mentioned that biosurfactant production has been traditionally
considered as a mechanism to enhance the biodegradation of hydrocarbons via pro-
moting their apparent aqueous solubility [429 438] or through promoting the inter-
action of microbial cell with hydrocarbons [435,439].
Biosurfactants would be appealing alternatives for classic chemically synthesized
surfactants conventionally used in petroleum industry. The main advantages of biosur-
factants over the chemically sensitized ones are being temperature tolerant, biodegrad-
able, general nontoxicity to humans, pH-resistant, possibility to be produced in-situ in
oil reservoirs, and being less expensive and more environmental friendly [22,31].
Moreover, biosurfactants can tolerate salt concentration as high as 10%, while only 2%
NaCl is enough to deactivate the synthetic conventional surfactants [441,442]. The
other appealing characteristic of biosurfactant making them more fruitful than